
#Iso 17025 2017 changes from 2005 iso
The second is important for laboratories already meeting ISO 9001 requirements for management systems it simply requires a 9001-compliant management system that meets all the relevant requirements of the new 17025. The first option is to comply with an explicit list of requirements, which broadly follow those in the 2005 edition. The most obvious change is that two broad options are now set out (labelled as options A and B). The big changes, however, are in the management system parts of the Standard. The standard also has a stronger focus on information technologies and (according to the ISO News release) “ … incorporates the use of computer systems, electronic records and the production of electronic results and reports”. Another, smaller, shift in technical emphasis is on sampling, where we see clearer more detail in the requirement for sampling and pre-treatment and a clearer set of factors to be recorded. Establishing clear decision rules is a key element of Eurachem’s guidance on the use of uncertainty in compliance assessment. Although the practical change is not large, we do notice an explicit requirement to document the decision rule used in conformity assessment.
#Iso 17025 2017 changes from 2005 full
We also see an emphasis on conformity assessment instead of the 2005 edition’s occasional reference to statements of compliance with specifications (for example in the reporting requirements), conformity assessment, too, has a detailed subclause on reporting statements of conformity and – unlike the 2005 edition – the detail applies in full to both testing and calibration certificates.
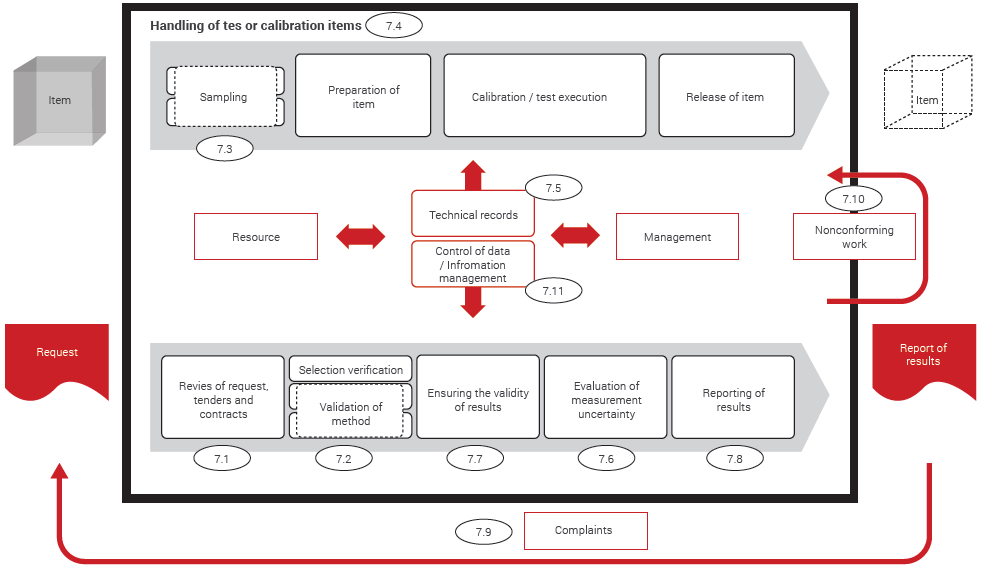
We’ll return to metrological traceability below. In 2017, metrological traceability has a whole new subclause of its own, tucked into Resource requirements, and a two-page informative Annex. In 17025:2005, the term ‘metrological traceability’ barely made an appearance metrological traceability was addressed by requirements for calibration. The most prominent example of this is the new emphasis on metrological traceability (see our Eurachem Guide on Traceability for more on this topic). What does seem to have changed is the emphasis on different elements. The distinction between reporting requirements for calibration and testing remains. Laboratories are still expected to train staff, to calibrate equipment properly, to validate their methods, and to evaluate and report measurement uncertainty where appropriate. The large difference in structure does not, however, signal great changes in most of the familiar technical elements of 17025. More recent CASCO standards have five sections: General requirements (which cover impartiality and confidentiality) Structural requirements, covering the nature and legal status of the organisation accredited Resource requirements, a substantial section dealing with personnel, facilities, laboratory environment and equipment issues Process requirements, which include contract review, many of the familiar method selection, validation, measurement uncertainty and reporting requirements and, finally, Management requirements. The 2005 edition was split into Management requirements and Technical requirements, appearing in that order. It has a new structure, closely aligned with all recent 17000-series standards from the CASCO stable. What can laboratories expect to see when they read the new Standard? We take a look.Īt first sight, the Standard looks completely different from the 2005 edition. The new version of ISO/IEC 17025 - General requirements for the competence of testing and calibration laboratories - has slipped in under the wire to become ISO/IEC 17025:2017.

